- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Vaccine Order, Distribution, Tracking, and Monitoring in Hong Kong Vaccination Program
- Home
- Back to Journal
- Article Details
Abstract
Hong Kong vaccination program launched at the end of February of 2021 to fight against the COVID-19 pandemic. The vaccine program is still ongoing and has been operated stably, with Hong Kong currently having more than 80 percent of people getting fully vaccinated. Since logistics of vaccine supply is critical to a successful program, in this work, we introduce our vaccine supply chain and explain the system of vaccine order and distribution, and the way tracking and monitoring by an online platform developed by the Logistics and Supply Chain MultiTech R&D Centre.
Keywords: Vaccine supply chain, Vaccine logistics, Information system, Hong Kong vaccination program, National immunization program, COVID-19
Introduction
Vaccination is an effective way to safeguard the public from COVID-19. Since the end of February 2021, the Hong Kong SAR government started to provide the public with two types of vaccines: (1) mRNA technology platform, by Fosun Pharma in collaboration with the German drug manufacturer BioNTech (hereafter referred to as BioNTech); and (2) Inactivated virus technology platform, by Sinovac Biotech (Hong Kong) Limited (hereafter referred to as Sinovac) [1]. In [2], the author explores the optimal method to distribute the limited supply of vaccines to the citizen in a way that matches the supply with demand and minimizes the cost, especially for the Hong Kong immunization program. Before the vaccination program kicked off, this work helped policymakers draw up the plan for the vaccination program by helping them have a more logical and optimized methodology to plan vaccine distribution over time.
However, the implementation of the vaccine program requires not only regulating the supply of vaccines, but also overall schemes such as planning the supply chain of vaccines, last-mile delivery, and operating vaccinations for CVCs. Especially, since the expiration deadline of the COVID-19 vaccines is tight [3], the efficient management of these is crucial for the success of the vaccination program [4]. Furthermore, the vaccines must be efficiently distributed so that they can reach the citizens without waste or bottlenecks in the supply chain. Therefore, it is essential to design a smooth and efficient vaccine distribution supply chain that minimizes wasted vaccines and enables prompt and accurate delivery and use of vaccines. In this work, therefore, we focus on this part as an effort for a successful vaccination program in Hong Kong.
For the Hong Kong vaccination program, Logistics and Supply Chain MultiTech R&D Centre (LSCM) has cooperated with the government to support vaccine programs in various ways. First, LSCM provided a system to monitor the vaccine delivery process. In particular, the e-Lock system developed by LSCM secures the transportation of the BioNTech vaccines from the warehouses to the Community Vaccination Centers (CVC). Also, LSCM offered a real-time dashboard to keep track of the usage of the vaccines in each CVC. Lastly, LSCM implemented an online platform for each CVC to request vaccines and to report vaccine usage, and for a Central Command Centre (CCC) to manage all CVCs requests and to place the final order to the warehouse. The information provided in the online platform enables us to monitor the inventory and decide how many vaccines should be ordered in each CVC daily.
In this work, we introduce the supply chain of the vaccine with each CVCs operation and the online platform, which is implemented in the Hong Kong vaccination program. This system enables to speed up and secure the delivery of the vaccines and enhance the efficiency of the vaccination program. Although the systems are provided separately for both vaccines, BioNTech and Sinovac, we only cover the BioNTech case in this manuscript. The logistic process of the BioNTech is much more complicated than that of Sinovac since it has multiple critical Use-By time points in its shelf life. This discussion will also help understand the handling of Sinovac.
We proceed with this exposition as follows. Section 2 introduces the vaccine supply chain design we have implemented in Hong Kong. In Section 3, we explain the vaccine delivery from the warehouse to each CVC with the process of vaccine order and fulfillment. Section 4 includes the operation at CVCs for inoculation to citizens and Section 5 explains our vaccine order algorithm based on a rolling horizon approach. In Section 6, we explain the information system with our online platform which records the necessary information and enables the staff to monitor the process. Section 7 shows the vaccine void rate in the Hong Kong vaccination program and classifies the rate by reason. Finally, we discuss all the above systems and conclude the work in Section 8.
Vaccine Supply Chain Design
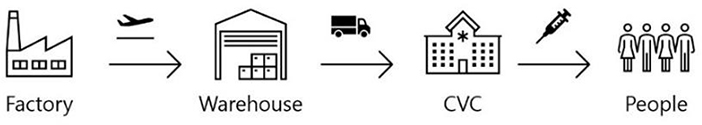
Figure 1: Vaccine distribution process.
In this section, we describe the vaccine supply chain of the vaccination program implemented in Hong Kong from a distribution point of view. Figure 1 illustrates a generic representation of the vaccine distribution process. The process consists of four echelons from the manufacturer to the end-user (i.e., the citizen): the factory where the vaccine is produced, the warehouse where the vaccine is stored, the CVCs where the inoculation is administered, and the citizen who are the recipients of the vaccine. The vaccine supply chain in our vaccination program involves not only the delivery but also the vaccination operation at each CVC and inventory management for the efficient distribution system. Therefore, in order to enhance the vaccination program successful with this supply chain, it is important to design the supply chain properly by considering how each part in the supply chain (delivery, operation, and inventory management) operates and how it is connected to each other to make the system seamless and efficient.
Since the supply chain must be tailored to the product, we should also consider the characteristics of the product when designing the supply chain. In the case of vaccination programs for sudden outbreaks like COVID-19, they have several unique characteristics such as high uncertainty in both supply and demand and the mass distribution under time pressure, which differentiate it from the supply chain of other products. In addition, the BioNTech vaccine which is one of the COVID-19 vaccines requires a special cold chain distribution with quite a short use-by time to be safely stored. Therefore, when designing the supply chain, we must consider the special conditions discussed as follows.
Requirements for BioNTech vaccine:
- The vaccine needs to be stored at ultra-low temperature (-70°C) before thawing.
- Once the vaccine is thawed, undiluted vaccine vials could be stored in the refrigerator with a temperature from +2°C to +8°C for up to 5 days (Since 16 September 2021, it has changed to 30 days [5]).
- The vaccine must be diluted within 2 hours in room temperature (up to 25°C).
- The vaccine must be used within 6 hours after dilution.
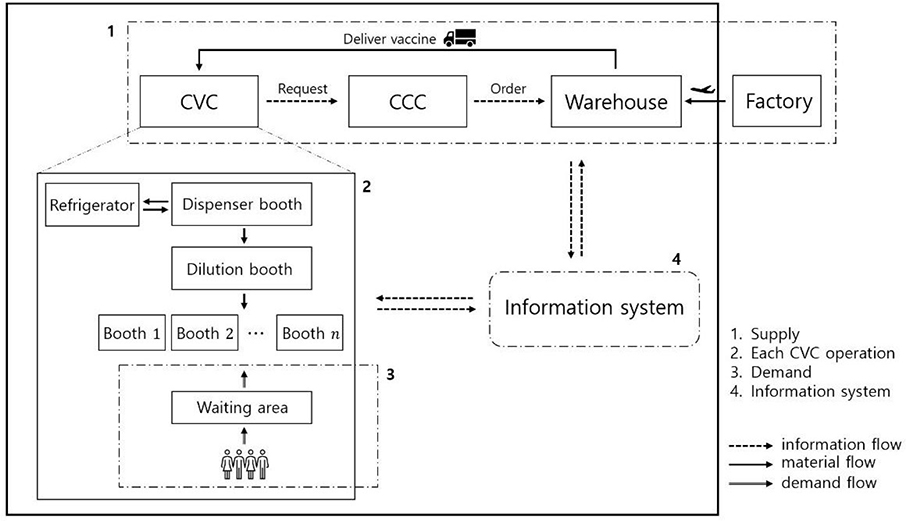
Figure 2: Illustration of vaccine supply chain design.
Taking these special characteristics of the BioNTech vaccine into account, we modeled the entire supply chain of vaccine logistics and operations and implemented it in practice in the Hong Kong vaccination program. Figure 2 shows this supply chain design. The first part of the supply chain is the delivery from the factory to a CVC. Since the delivery process from the factory to the warehouse is under the responsibility of the vaccine manufacturer, we only deal with the delivery process except for this part to discuss the vaccine distribution process for Hong Kong. The arrows indicated by dotted lines represent the flow of information such as the quantity of vaccine they need or order while the solid line indicates the flow of materials which are shipped. Specifically, the information about the quantity of vaccine orders are transferred from each CVC to the warehouse after being confirmed by the CCC and the vaccines are delivered from the warehouse to each CVC respectively. The second part of Figure 2 is the distribution process within each CVC. After the vaccines are shipped from the warehouse to each CVC, the CVC dispenser stores the vaccines in the refrigerator with a checking process and distributes the required vaccine vials to the dilution team. Then, the dilution team staff reconstitutes the vaccine with diluent and prepares the syringes. The syringes are then distributed to each inoculation booth for injection to citizens. The end-user of the vaccine is the citizens who get inoculated, so there is a demand flow which is represented in the third square in Figure 2. Citizens go to the CVC where they make appointments to get vaccinated. When they arrive at the CVC, they go to a waiting area of the CVC and go get inoculated in turn. Lastly, all the procedures are recorded and managed via an information system for effective vaccine management. The online platform designed by LSCM enables the relevant users to monitor the status of their vaccine stocks and orders and the flow of the vaccines.
Vaccine Order, Fulfillment, and Delivery
In this section, we describe the process of fulfilling and delivering the vaccine once it is ordered. As mentioned in the previous section, the BioNTech vaccine has special temperature conditions and short shelf life for safe storage after being thawed or diluted. Therefore, the delivery system for this vaccine and the additional processes involved pose greater challenges than other delivery systems. First, most healthcare providers have no ultra-cold freezer that meets the temperature requirements for the storage of the BioNTech vaccine, while the vaccine should be stored at a proper place having the special freezer for long-term storage and distributed to all CVCs from this place upon orders. To solve this problem, the Hong Kong government outsources the storage of vaccines and transportation to each CVC to a private company by designating DCH Auriga (DCHA) as the exclusive distributor of the vaccines for the Hong Kong vaccination program [6]. To increase the effectiveness of vaccine supply chains, WHO recommends outsourcing certain functions of the supply chain to another private sector [5,7]. Indeed, several research studies show that such outsourcing is beneficial [8, 9, 10]. The DCHA is one of the largest healthcare distributors in Hong Kong and has strong experience in managing sophisticated pharmaceutical supply chains with its cold chain expertise. By outsourcing the storage and delivery of the vaccine, therefore, the whole supply chain could be smoothly and effectively managed. Second, for the successful introduction and implementation of the BioNTech vaccine, we must tackle the challenge of convincing the citizens of Hong Kong that the delivery process of the vaccine is safe. Since the BioNTech vaccine which is one of the novel mRNA vaccines has unusually delicate requirements to maintain its efficacy, the citizens could have hesitancy against getting vaccinated and have doubt in the safety of its supply chain management. This problem is mitigated by recording the information about the safety of vaccine delivery through an information system, which will be discussed in Section 6. By tracking and monitoring the delivery of vaccines on the information system, it is ensured that the vaccines are delivered and managed in a proper condition safely. As a result, it helps the recipients of the vaccine trust the vaccine management system.
Vaccine Order
Figure 3: The flow of vaccine order.
The flow of vaccine order is shown in Figure 3. Each CVC requests the necessary quantity of vaccines based on its inoculation booking number and vaccine inventory amount around 2 p.m. in each day. After all CVCs placed requests, the CCC pharmacist manages the requests of all CVCs to coordinate and confirm the requests. And then the CCC pharmacist places the final order for all CVCs around 3 p.m. Then, according to the confirmed final order from the CCC, the delivery staff prepares to deliver the vaccines from the warehouse to each CVC 2 days after the order is placed. A detailed explanation of the order logic or the online vaccine ordering platform is deferred to Section 5 and Section 6, respectively.
Order Fulfillment
The original factory supply of the BioNTech vaccine is in a carton containing 195 vials, which is called a “pizza box” [11]. After the vaccine is shipped from the factory to the warehouse, the pizza boxes are stored in the ultra-cold freezer without repackaging it. Once the vaccine is thawed, it could be stored for up to 5 days in the refrigerator with a temperature from +2°C to +8°C. Hence, we should make the delivery process prompt and systematic to maximize the period of storage at each CVC after the delivery.
Upon receiving the final order from the CCC around 3 p.m., the DCHA staff prepares for the delivery by taking the pizza boxes as much as the required quantity out of the freezer and thawing the vaccine overnight. Note that once a carton of vaccine is opened, all vaccine vials in the carton must be thawed immediately. Thus, the total amount of the ordered vaccines from all CVCs each day must be in the multiple of 195 vials to match it with the number of all vials in the pizza boxes taken out of the freezer. On the next day, the thawed vaccines are repackaged as one of the three types of packages (195 vials, 5 vials, and 1 vial per box) and arranged to be delivered the day after. Then, the prepared vaccines are delivered the next day early in the morning, enabling the CVCs to start vaccination operation from 8 a.m. after the process of checking the delivered vaccines.
Order Delivery
Figure 4: Safe delivery with e-Lock system.
In the delivery process, the e-Lock system developed by LSCM is applied to the delivery system for real-time tracking and monitoring, to make sure the vaccines are delivered in the uncontaminated state safely in time. The e-Lock system registers bundled information of e-Lock ID, handheld device ID, departure location, and destination location. The delivery process from the warehouse to each CVC is as follows. The truck driver loads the vaccines on the truck and locks the truck door with the e-Lock and the mechanical key as shown in Figure 4. Then the driver activates the e-Lock with handheld and transports the vaccines to CVC. The truck’s GPS and the e-Lock status information are monitored on the console panel of the e-Lock monitoring platform. When the truck arrives at its destination, the driver deactivates the e-Lock with the handheld device to end the secured trip. The driver unlocks the e-Lock with the mechanical key and hands over the vaccines to CVC. If an abnormality such as e-Lock is tampered with or geofencing is violated during this process, an alert will be triggered.
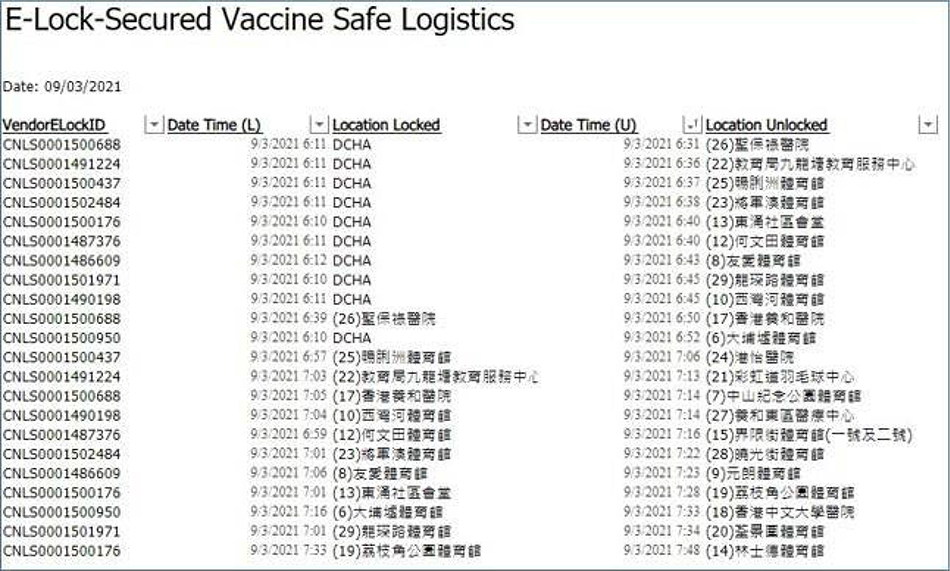
As you can see in Figure 5, the information stored through the e-Lock system allows you to check in real-time the locked location before delivery, the unlocked location after delivery, and the arrival time of each CVC. For example, let us look at the delivery process of a delivery staff whose VendorELockID is CNLS0001500688. In the first and tenth rows of Figure 5, you can check the delivery route of the staff with this ID. On March 9, 2021, this staff loaded the necessary vaccines from the DCHA warehouse and locked the truck at 6:11 a.m., and at 6:31 a.m. opened the truck door at CVC No. 26 to unload the vaccines. In addition, the truck door was locked again at 6:39 a.m. at CVC No. 26 and the truck continued the delivery process by heading to CVC No. 17. Lastly, it can be seen that the truck door was opened at CVC No. 17 at 6:50 a.m. for unloading the vaccines there.
Figure 5: Safe e-Lock secured logistics.
CVC Vaccination Operation
After the vaccines are shipped from the warehouse to each CVC, the CVC dispenser team receives the vaccines with a check-in process and stores them in the refrigerator. During the CVC working hours, the CVC dispenser team takes the required amount of vaccine from the refrigerator to the dilution team for them to dilute the vaccine and prepare syringes. Then, the dilution team distributes the syringes to the inoculation team of each booth for inoculation. The successful inoculation numbers, together with the voided dose (wastage), are also reported back in real-time so that we can know the consumption rate of the syringes prepared.
Since the precise handover of vaccines between each team is the cornerstone of a successful vaccine program, loggings with vaccine batch registration information (what), handover time stamps (when) and the relevant staff cards (who) are recorded in each handover procedure. In addition, the verification process automatically records the delivery process in real-time on the online platform and the users enable tracking and monitoring in real-time. Therefore, it is possible to oversee the delivery, inventory control, and order planning of the vaccines with a real-time vaccine management system. Now, we describe each process in detail.
Receiving process:
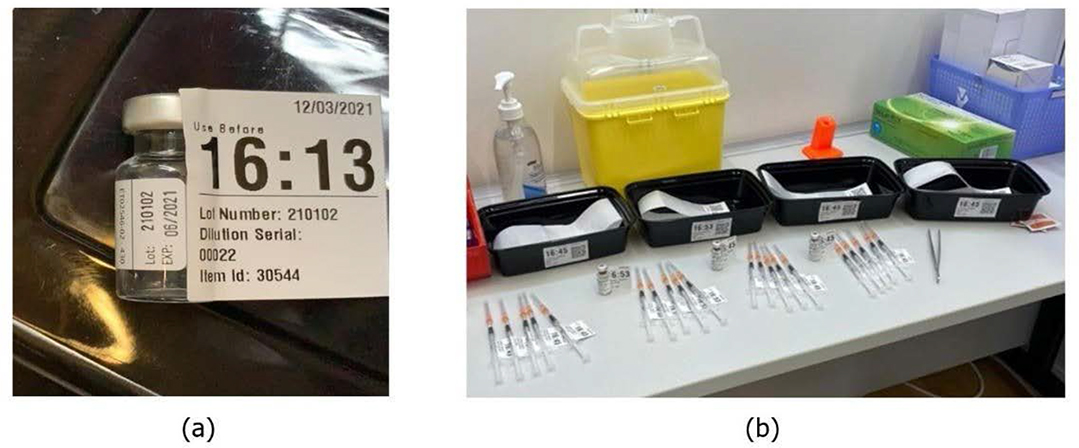
When the delivery staff arrives at a CVC, the CVC pharmacist scans the personnel NFC card of the driver and verifies the number of vials and saline received, confirming delivery of goods. Also, the CVC dispenser checks the vaccines and saline individually by reading the barcode in each vaccine package like in Figure 6 (a). The barcode tells the information that includes the vaccine quantity, lot number, thawed timestamp, and use-before timestamp. The vaccine information is automatically recorded on the online platform at the time the barcode is read. Then the vaccine will be put in the fridge waiting to be used.
Figure 6: (a) CVC check-in (b) Dispensing
Dispensing process:
According to the booking number of people for inoculation in each time slot and the information of the real-time waiting area’s queuing line, the dispenser transfers the required quantity of vaccines to the dilution team to prepare syringes to be used. At this time, the vaccine is brought out to be dispensed in the order of the shortest shelf life (the earliest thawed time) among the total remaining stock according to the rule of first expired first out (FEFO). The dispenser prints two dispensing labels for the dilution team, which has the information: the dispensing timestamp of the diluent saline and vaccine, the quantity of them, the thawed timestamp of the vaccine, and the use-before timestamp of the vaccine (within two hours at the room temperature), as shown in Figure 6 (b). If the vaccine vial is found abnormal, such as having dirt or having a loose cap, the whole vial will be discarded with reason recorded.
Dilution and syringes preparation process:
After the dilution team takes over vaccines and diluent saline, they start the dilution process and print nine label stickers per vial, with 1 for syringe container tray, 1 for worksheet, 1 for the vial bottle, and the others for 6 syringes, as shown in Figure 7. The information on the stickers is all identical and includes the assigned dilution serial number, vaccine lot number, and the use-before timestamp (within 6 hours after dilution). So, each syringe is trackable back to the original vaccine vial, i.e., in case any anomalies arise, the problematic vaccine vial and lot number can be traced back. Now, the trays with syringes are distributed to the inoculation booths, and the system will associate the information of the syringe container and the inoculation booth representative during the handover of the syringes. The booth representative returns the used empty vial bottle and the syringe tray to the dispenser. If there are vaccine syringes that need to be discarded, they are placed on the tray at the booth and delivered back to the CVC dispenser for a calculation of the waste volume. The voided syringes are then recorded in the system in dose units along with the reason.
Stock taking and vaccine ordering:
For each CVC, the CVC dispenser requests the needed quantity of vaccine and diluent saline through the LSCM online platform. Vaccine requests must be submitted to the CCC side between 2:00 p.m. to 2:20 p.m. after confirming the vaccine and saline inventory. The ordering system on the online platform, which has the information related to demand and supply, generates the CVCs’ order requests for vaccines with the unit of a pack of 195 or 5 vials and for saline with the unit of a pack of 20 ampoules. The order request of each CVC is automatically calculated on the system based on the CVC’s inoculation appointments information, which is updated daily, and current inventory information. For the inventory information, the dispensing staff performs inventory checking and makes sure that the computer record on the online platform matches with the physical stocks. Figure 8 shows the vaccine stock in the refrigerator.
Figure 7: Dilution label sticker for the vial (b) Vaccine trays and syringes with label stickers.
Figure 8: Vaccine stock.
If there is any CVC that misses vaccine request till 2:20 p.m., the CCC staff would call the CVC staff to make them submit the request immediately. After all CVCs placed the requests, the system automatically rounds up the total vaccine requested from all CVCs to fit multiples of 195 vials which is the thawing unit, and the odd vials are distributed to the CVCs with Hamilton’s method (2 See reference [12] for the explanation of Hamilton’s method). After this automatic adjustment, the CCC pharmacist places the final order. Note that the CCC staff may adjust the final vaccine and diluents quantities assigned to each CVC as needed, so the final order may differ from the original request from CVCs. The CCC user then submits the final order by 2:45 p.m. and each CVC user would be able to view the adjusted order after 2:45 p.m. Once the final order is placed, the ordered quantity of vaccine and diluents will be delivered two days after the date of application. A more detailed explanation of vaccine ordering logic is described in the next section.
Vaccine Order Algorithm Based on a Rolling Horizon Approach
Figure 9: Illustration of rolling horizon planning of vaccine order request.
In a COVID-19 Vaccine supply chain; there are several conditions to consider when ordering to ensure adequate stock of vaccines at each CVC. First, the special requirement for the shelf life of the vaccine must be taken into account. Once the vaccine is thawed and delivered to each CVC, it can be stored in the refrigerator for up to 5 days. Therefore, we should not allow our inventory to remain unused for more than 5 days. Second, we should consider that the citizens who made appointments but could not come (no-show) or changed their appointments. In the Hong Kong vaccination program, the citizens can only make new appointments on an online platform two days prior to the inoculation date. Therefore, the number of appointments within 2 days should be discounted (with no-show or cancelled/changed appointments), after 2 days should be on the rise (with expected new bookings). If these are not properly reflected when deciding the quantity of the order, there could be a possibility that a large number of vaccines are voided without being used within the shelf-life period, or this may be a factor that interferes with scheduled inoculation due to insufficient quantity. Therefore, to avoid wasting vaccines, we use a reactive approach in the context of a rolling horizon approach that updates demand and inventory on a daily basis, ensuring that order volumes are properly re-planned as needed.
In Figure 9, two consecutive iterations of the rolling horizon planning are shown. For notational convenience, we refer to T as the date of placing the order of the first of the above two iterations. Then, the next day is T+1 which is the start of the second rolling horizon. In the rolling horizon planning for the vaccine order request system, the time horizon required to calculate the quantity to be ordered on day T is from the time of ordering in day T today T+3. This span of the period is divided into the frozen interval containing the information of the days T, T+1, and T+2, and the free interval having the information of the day T+3. On day T, the appointments on the days T, T+1, and T+2 are not open for new bookings, and we will consider the actual booking number with a discount rate, i.e., to take into the factor of no-show and appointment cancellation. On the day of the order T, especially, the number of undispensed vaccine vials and the remaining number of appointments are considered as the current stock and the remaining demand of day T, respectively. Therefore, the current remaining stock and the current remaining demand are taken into account in calculating the order quantity, excluding the vaccine usage that has already been realized at the time point of ordering. On the other hand, the appointments of the day T+3 in the free interval are subject to change on day T on the online booking system, the demand of T+3 is considered as safety stock. We will consider the actual booking rate on T+3 with an expectant rate, i.e., to take into account of appointments variations expected on T+3. The order requests from each CVC after these calculations take place around 2:00 p.m. on a daily basis. The order on day T is for the delivery of day T+2. Orders on Friday are subject to slight changes to account for shipping holidays on Sunday, but the order logic is not significantly different, so we will omit the description of it.
When a new planning period starts, we confirm that the part that was the free interval in the previous horizon is frozen, the number of appointments of the next day is updated with new demand data, and the realized data is updated, so you can apply more accurate information to the calculation. This process continues repeatedly until the vaccination program is complete. The implementation of this algorithm considers the uncertainty of the COVID-19 vaccine supply chain associated with the difference between the planned and the actual appointments. Thus, the calculated ordering quantity covers such uncertainties by constantly applying the current state of the demand and supply. That is, the ordering system is quickly adjusted to include new information about the quantity used on the day and the new demand data as safety stock; thereby it improves the accuracy of the calculation. As a result, this planning makes the vaccination program more successful by minimizing the amount of vaccine waste. In addition, all the calculation is automatically done on the online platform, making the system efficient and convenient. The detailed order logic is as follows.
Assume that the order date is T. The supply is the current stock inventory except for the vaccine which has already been dispensed at the time of the request (which include the order delivery on the current day T, as it will be received before 7:00 a.m.), plus the amount that is scheduled to be delivered on the next day T+1. On the other hand, when calculating demand, we take into account the number of appointments from the day T to T+3. Apart from the values for T, T+1, and T+2, the value for T+3 is for safety stock. Note that there is a requirement for one-day safety stock in each CVC. In Hong Kong, the booking appointments for BioNTech are frozen 2 days before the inoculation date, so the number of appointments for T+3 is subject to change, we consider the expectant rate in anticipation of further increase or decrease. In addition, the original number of appointments is adjusted to the number of discounted bookings. Originally, the discounted booking rate was set to less than 100% in consideration of no-shows, but now it is set to 105% as shown in Figure 10 considering the walk-in quota. In addition, if adjustments are required due to outreach programs or other reasons, the required quantity is indicated in the Adjustments row.
Finally, the demand is the sum of the discounted booking numbers of T, T+1, and T+2, the discounted booking number of T+3 considering the expectant rate, and all adjustments T, T+1, and T+2. Then, the difference between the demand and the supply is the automatically calculated number of order requests for the CVCs.
Information System with Online Dashboard
In this section, we explain our information system which connects the whole supply chain and provides proper information to the relevant staff. Information systems in the vaccine supply chain enable better and more timely decision-making to the staff in the vaccination program and make the supply chain smooth and efficient [13]. Also, for the effective functioning of the vaccination program, [14] puts an emphasis on the existence of information systems in the vaccine supply chain. Therefore, the design of the information system, as well as its existence, is important factors in the success of the vaccination program.
The vaccination program in Hong Kong is managed through the secure online platform developed by LSCM. The information needed for each CVC and the CCC along the whole supply chain is collected and recorded by the relevant staff at each stage of the supply chain at the right time in the process. In addition, the information is automatically recorded to the platform by reading barcodes of the vaccines and their relative products, and the dashboard provided on the online platform shows the information that is calculated automatically from the relevant numbers. Thus, this online platform helps the vaccination program to be operated seamlessly and efficiently by providing accurate and timely information to the relevant staff conveniently. Now, we describe the detailed functions of the online platform.
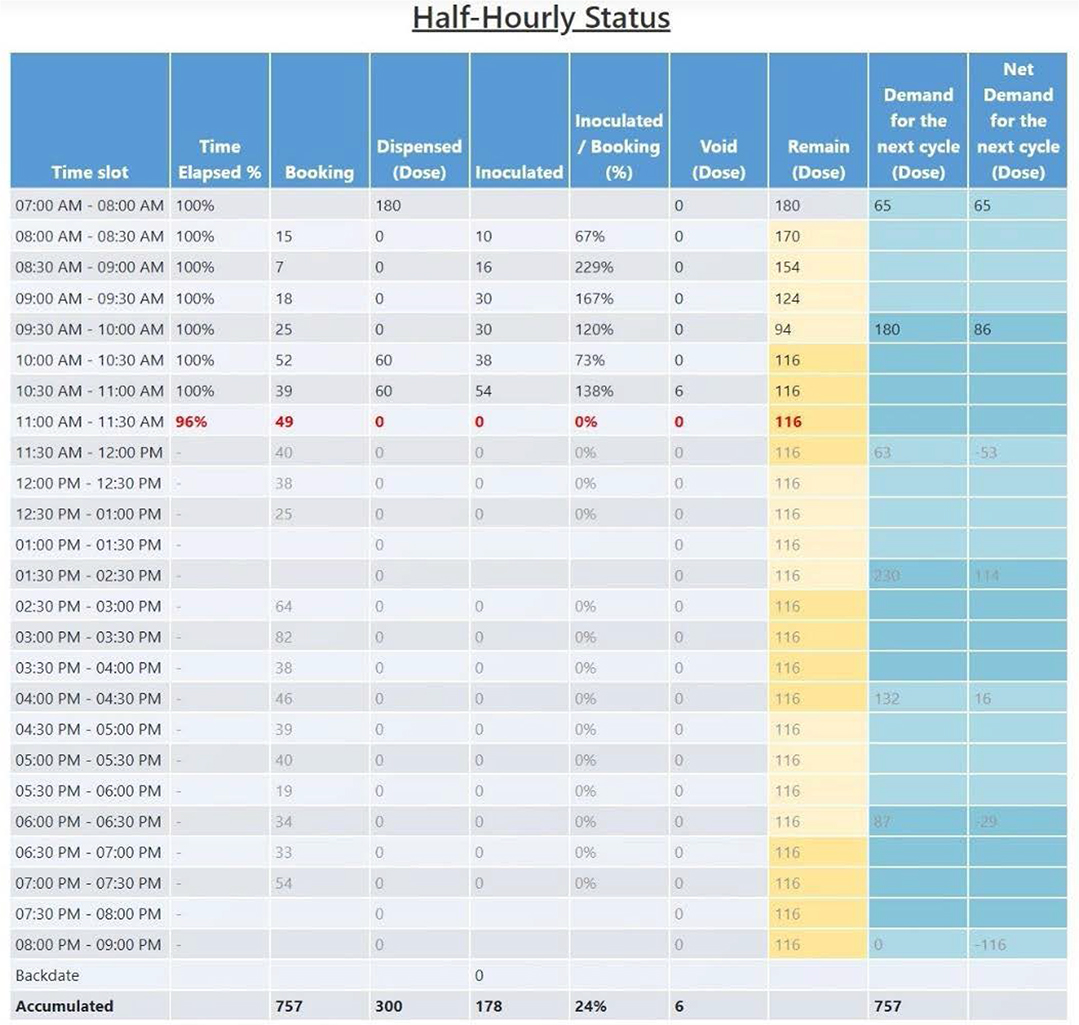
Figure 10: Half-hourly status during the day.
Dashboard functions for CVC
Each CVC is a place where vaccination is operated, and its main tasks are to order vaccines, receive the delivered vaccines, manage inventory, and perform vaccinations. In doing these tasks, it is required for each CVC to record, check, and monitors each process thoroughly for the successful implementation of the vaccine program. To facilitate the tasks of CVCs, the dashboard provides several reporting functions for each CVC including the following basic functions: half-hourly status, daily activities, and booth activity log.
As you can see in Figure 10, on the half-hourly status page, the number of appointments, the number of inoculations, and the ratio of the two are indicated in each time slot. The time slots for the available vaccination time (from 8:00 a.m. to 8:00 p.m.) of the day are divided into 23 slots at 30-minute intervals except for the one-hour time slot from 1:30 p.m. to 2:30 p.m. considering a lunch break from 1:00 p.m. to 2:30 p.m. In addition, one hour before and after the vaccination operation time is the preparation time (from 7:00 a.m. to 8:00 a.m.) and finishing time (from 8:00 p.m. to 9:00 p.m.) of CVC, respectively. In this page, not only the above numbers but also the amounts of vaccine distributed, discarded, and remained are recorded in dose units by this time slot. All the numbers are recorded in real-time, so you can monitor the performance of the process for all CVCs in real-time, which enables the relevant staff to utilize the information in a timely manner. For example, if the current time slot is from 11:00 a.m. to 11:30 a.m. as shown in Figure 10, the information of the present time slot is displayed in bold red text. Also, the accumulated information of the day can be checked like at the bottom line in Figure 10.
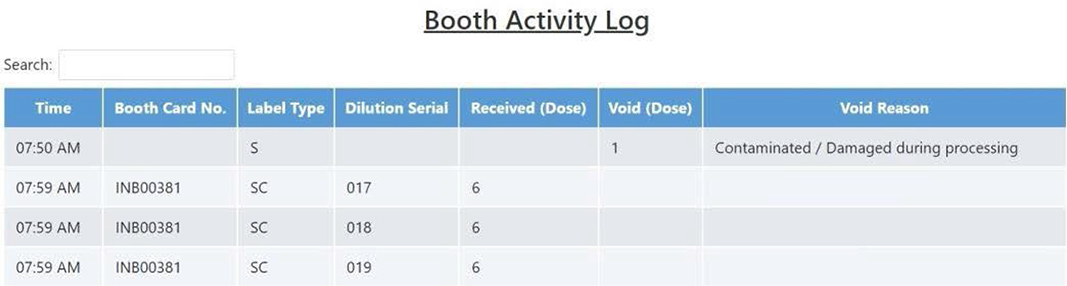
Figure 11: Booth activity log.
On the daily activity page, you can monitor the vaccine usage and inventory for the day, and the discarded vaccines are recorded by reason. In addition, the daily activity logs for the quantities and lot numbers of vaccines delivered, dispensed, discarded, and stocked during the day are recorded down to the minute. The booth activity log page, on the other hand, records the syringe usage information of each inoculation booth, which includes booth number (or booth card number), the number of syringes received with the dilution serial, and the timestamp of syringes received or voided. An example is shown in figure 11.
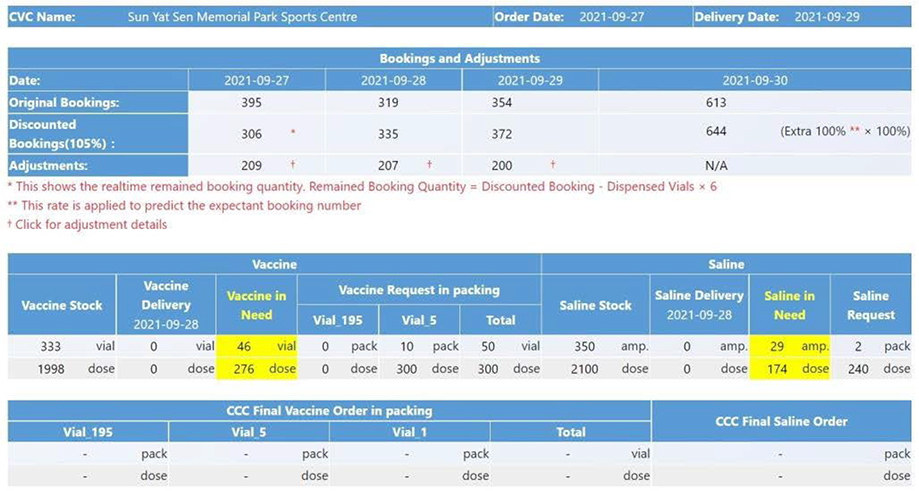
Figure 12: The BioNTech vaccine request page for a CVC.
There is also a page where each CVC places vaccine request, which is shown in figure 12. The CVC dispenser uses this form to place the vaccine and saline request. Before submitting the request, the CVC dispenser should make sure the vaccine and saline inventory is correct and refresh the screen to view the most updated booking information and inventory information. The order request is then automatically calculated based on the numbers of bookings, stock inventory, and expected delivery of the next day. As we explained in the prior section, there is order logic to calculate the request number in these numbers, so the logic is reflected and implemented on this page. As a result, the needed vaccine and saline quantities are represented automatically once the user confirms the remaining inventory like in the yellow part of
figure 12.
Dashboard functions for CCC
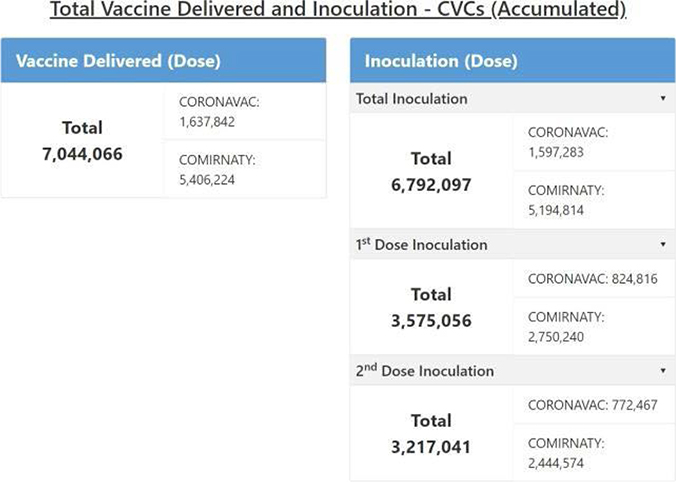
The CCC is responsible for monitoring vaccine usage and inventory in all CVCs, for finalizing vaccine requests from CVCs, and for placing orders. Therefore, CCC users can access all dashboard functions for each CVC. In addition, there is an extra CCC overview page, where you can see the total accumulated number of vaccines that are delivered and inoculated by vaccine type as shown in Figure 13.
Figure 13: Total vaccine delivered and inoculated in all CVCs.
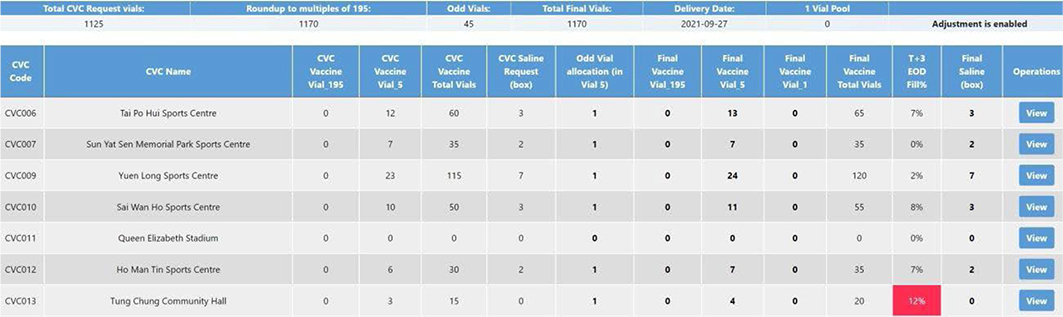
Figure 14: CCC BioNTech vaccine order placing.
Also, the CCC user can manage the vaccine order requests from all CVCs on the order placing page as shown in Figure 14. After all CVC requests have been submitted, the orders from the CCC side will be automatically recalculated to make the final total vaccine vials a multiple of 195. As mentioned in the previous section, the total quantity of the vaccine vials requested from all CVCs should be a multiple of 195 vials, so it is rounded up to a multiple of 195 to be distributed. For example, in the first row of Figure 14, you can see the total requested vials from all CVCs are 1125, and the number rounding up this is 1170. The remaining 45 vials are allocated to several CVCs as additional stock. As shown in Figure 14, the CCC user can check the requests of CVCs after the automatic adjustment and the order of the CCC after the remaining vial allocation on this page. The CCC pharmacist can further adjust the final vaccine/saline quantity allocated to each CVC if needed. After all the adjustments are done, the CCC pharmacist can submit the final orders.
Vaccine Waste in our Vaccination Program
Given the stringent storage requirements of the BioNTech vaccine such as several tricky storage temperature requirements or short shelf life at room temperature, the rate of COVID-19 vaccine wastage was anticipated much higher than other vaccines’ discard rates. Actually, when the Hong Kong government was preparing before launching the vaccination program, officials expected the disposal rate of the COVID-19 vaccine to reach about 5%. Nonetheless, the vaccine discard rate in the vaccination program in Hong Kong has shown much lower than expected earlier. From March 6, the beginning of the vaccination program, to the end of November, the total amount of dispensed vaccine is 5,920,302 doses and the total vaccine discard is 33,611 doses, which results in the vaccine waste rate is around 0.57%. This rate is a considerably low percentage, almost one-tenth of the expected rate of 5%. Achieving these results would have required more effort in this program than in any other immunization programs. And the rigorousness and efficiency of the distribution supply chain and CVC operations described in the previous section are included in this effort.
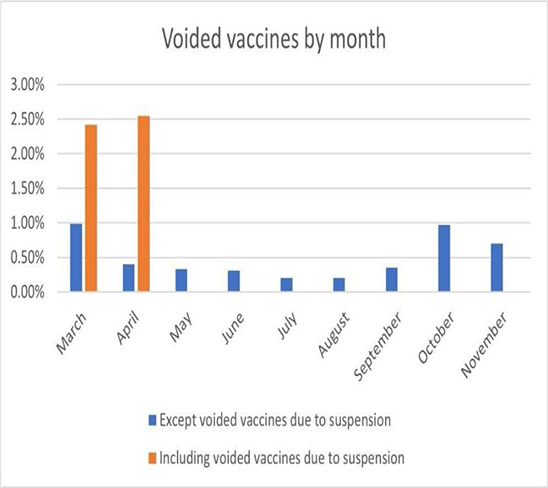
As can be seen in Figure 15, over the past nine months, vaccine discard rates were less than 0.50% for all but three months. Especially, in July and August, the rates are only 0.20% each, which is a remarkable result. In October and November, the discard rates show a bit higher than the previous months, but still low figures as of less than 1.0%. The March and April categories in Figure 15 each have orange bars separately from the blue bars, both of which represent close to 2.5% vaccine discard rates. These figures include the number of vaccines that were discarded due to a temporary suspension of the vaccine program due to a packaging problem with the vaccine at the end of March. On March 24, 2021, after receiving a report that there was a problem with the vaccine packaging, vaccines with the same lot number were collected back for investigation. The immunization had been suspended from that day to early April and was resumed afterward. This happening is not common in the vaccination program (actually happened once) and the number of discarded vaccines due to this problem was considerable. Therefore, to distinguish this case, we provide the total discard rates including the number of the discarded vaccine due to the vaccine program suspension separately in orange bars. The figures excluding the number of vaccines discarded for this reason were marked with blue bars. All other months except March and April provide only blue bars because there were no such problems.
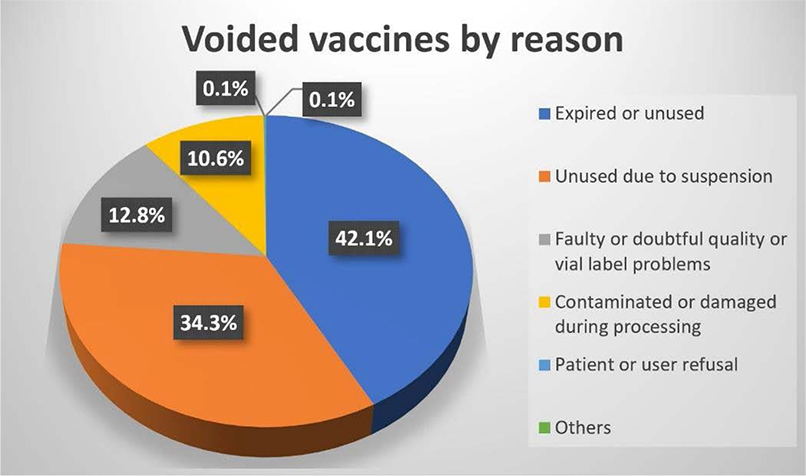
Then, what percentage of the total vaccine discard volume will be due to this suspension of the vaccine program problems? In Figure 16, the percentage of vaccine waste for each reason is shown as a pie graph. The amount of the voided vaccines due to the suspension accounts for 34.3% of the total amount. It is quite a sizable portion of the total vaccine waste. However, the largest proportion of the discarded vaccines was due to the expiration or unused. This category includes the case where the vaccine has expired on the shelf. Also included in this category are vaccines that were prepared in advance but were discarded because the session was ended. In addition, the reasons for discarding the vaccine include problems with the vaccine itself, problems with contamination during preparation or inoculation of the vaccine, patient or user refusal, and others.
Figure 15: Voided vaccines by month.
Figure 16: Voided vaccines by reason.
Except for the discarding of vaccines due to exceptional circumstances, such as when the manufacturer recalled the vaccines for investigation which resulted in the suspension of the vaccine program, most of the causes of vaccine discard fall into the following three categories: (1) Problems with the vaccine product itself, (2) negligence of medical staff performing vaccination and vaccine reconstitution, and (3) failure to use the vaccine due to the expiration date being passed. Particularly, the last one is the largest reason (42.1%) for discarding vaccines as we mentioned earlier. The rigorousness and efficiency of the vaccination operation in CVCs including the vaccine ordering system has helped to significantly reduce the number of vaccines that are discarded especially due to this reason which accounts for this substantial proportion. Therefore, it can be said that it is an important way for minimizing vaccine waste to properly understand the characteristics of the COVID-19 vaccine (BioNTech) and to have a thorough system for vaccine delivery, storage, and operation.
Discussion
We designed and introduced the vaccine management system with fast and safe delivery processes and thorough checking and recording in each step, which considered the characteristics of the vaccine with tight expiration dates and vulnerability. By introducing an e-Lock system for secure delivery and labeling functions with vaccine information for vaccine management, the entire process has been semi-automated with the use of handheld devices and personnel NFC cards, and an efficient and seamless system was created. Also, it has been possible to ensure the stability of the vaccine by thoroughly managing the shelf life of the vaccine, and tracing back when a problem occurs. In addition, each CVC’s daily vaccine orders have been determined using a rolling horizon approach, enabling vaccine inventory management using more accurate information updated on a daily basis. Particularly, we help all processes to be connected and flow more seamlessly by building an information system so that all data can be utilized through recording and tracking. By providing online dashboards that include relevant information to CVC and CCC managers and related parties on the online platform.
By making the above efforts, we have solved several difficulties in the COVID-19 vaccine supply chain. The COVID-19 vaccine supply chain differs from the existing vaccine supply chains in that it is a pandemic situation and the vaccine is a newly developed. In [15], the authors present a total of 15 challenges in the vaccine supply chain for this new type of vaccine. These are mainly categorized into manufacturing challenges, behavioral challenges, last-mile delivery challenges, cold chain challenges, and organizational challenges. Aside from the manufacturing challenges, which are not of topic for our work, we tackled the rest of the challenges. The following is how we have solved the rest of the challenges:
- The first challenge was to identify and design the vaccine supply chain tailored to the characteristics of the vaccine with the goal to minimize vaccine waste and to reach as many citizens as possible.
- Second, we solved the cold chain challenges, which are the major difficulty of the COVID-19 vaccine supply chain, by outsourcing the storage and delivering part to the private sector having the cold chain expertise.
- Third, we introduced an information system to solve the organizational problems of the vaccine supply chain. The information system helps the communication between related parties easier for the quick response, ensuring rapid vaccine distribution, central management of the vaccine, and transparency of vaccine programs.
- Also, by applying the appropriate technology to the supply chain, the system’s efficiency and stability, and furthermore, citizens’ anxiety about vaccination has been solved. The automatic system due to the technology gives citizens trust in the government’s vaccine program by ensuring the stability of vaccine management and CVC’s operation.
- Lastly, we enable the sustainability of the vaccine supply chain. By enabling the application of changing vaccine information and vaccine programs to the aforementioned technologies and information systems, it is possible to respond flexibly to the changes.
As a result of these efforts, we recorded the low vaccine wastage rate (0.57%) shown in the previous section and played a role in leading the vaccination rate of over 70% in Hong Kong. Amid this COVID-19 pandemic, these swift responses and efforts have paid off, but there are clearly areas that need to go further in addition to vaccine distribution. First, stronger training for healthcare workers working in vaccination centers is needed. As shown in Figure 16, there are many cases where vaccine waste is generated due to errors or mistakes in the process of vaccination execution and vaccine reconstitution. In addition, the success of the vaccination program requires not only the efforts in logistics and operation, but also the efforts of both the government and citizens. It is necessary to encourage vaccination based on a logical report on vaccination. In order to boost the current slow vaccination rate, cooperation and efforts in all aspects as well as operational aspects are important.
Conclusion
This paper explains the vaccine ordering and distribution system and its corresponding information system with the online platform applied to Hong Kong’s vaccination program against the COVID-19 pandemic. Since the cost of the vaccine itself accounts for a significant portion of the cost of immunization programs, it is important to minimize vaccine waste to avoid jeopardizing vaccination. In order to do this, we developed and introduced several devices to make the distribution channel flow seamlessly and designed the supply chain drawn from the understanding of the characteristics of the vaccine. In addition, since citizens’ anxiety about the new type of mRNA vaccine for COVID-19 has been able to become an obstacle to the vaccination program, to make the citizens rest assured for getting the vaccine, we have secured the stability of the overall process: the CVC operation, the storage, management, and distribution of the vaccine.
To the best of our knowledge, this is the first work to describe the system of the vaccination program in Hong Kong that is being implemented and to show the COVID-19 vaccine waste rate from the real-world data. The system presented here shows the efficient and well-organized distribution processes and CVC operations, and the corresponding results, which are represented by the figures about the amount of vaccine discarded, support this. Depending on how the vaccine distribution and inventory management and vaccination are operated, the vaccine discard rate can be significantly lowered or higher. Therefore, the system we provide will be of great help to countries that have not yet performed mass vaccination or countries that do but do not properly manage them. Or it will help prepare new vaccination programs for another new type of pandemic before it comes. We hope that the description of our system will increase the understanding of effective vaccine management.
References
- The Government of the Hong Kong Special Administrative Region, “About the Vaccines”. [View]
- Cheng CH (2021) Supply Driven Planning for a Vaccination Program in Hong Kong: An Optimization Approach. COVID-19 Pandemic: Case Studies & Opinions 02(01): 196–2021. [View]
- https://www.who.int/publications/i/item/WHO-2019-nCoV-vaccines-SAGE_recommendation-BNT162b2-2021.1
- https://apps.who.int/iris/bitstream/handle/10665/336603/WHO-2019- nCoV-Vaccine_deployment-2020.1-eng.pdf
- https://apps.who.int/iris/handle/10665/343080
- https://www.dchauriga.com/en/news-and-insights/dch-auriga-selected-as-the-logistics-partner-to-distribute-biontech-covid-19-vaccine-in-hong-kong/
- WHO, UNICEF, GAVI, Alliance, PATH. Effective vaccine management initiative: background. Geneva: WHO; 2010. V1. P. 7.
- Lydon, T. Raubenheimer, M. Arnot-Krüger, M. Zaffran (2015) Outsourcing vaccine logistics to the private sector: The evidence and lessons learned from the Western Cape Province in South-Africa. Vaccine, 33: 3429-3434. [View]
- USAID | DELIVER PROJECT (2010) Emerging Trends in Supply Chain Management: Outsourcing Public Health Logistics in Developing Countries. [View]
- Riewpaiboon, C. Sooksriwong, N. Chaiyakunapruk, P. Tharmaphornpilas, S. Techathawat, K. Rookkapan, A. Rasdjarmrearnsook, and C. Suraratdecha (2015) Optimizing national immunization program supply chain management in Thailand: an economic analysis. Public Health 129: 899-906. [View]
- Tinglong Dai, Jing-Sheng Song (2021) Transforming COVID-19 vaccines into vaccination. Health Care Manag Sci 24: 455–459. [View]
- https://math.libretexts.org/Bookshelves/Applied_Mathematics/Math_in_Society_(Lippman)/04%3A_Apport ionment/4.02%3A_Hamiltons_Method
- Michel Zaffran, JosVandelaer, DebraKristensen, Bjørn Melgaard, Prashant Yadav, K.O. Antwi-Agyei, and Heidi Lasher (2013) The imperative for stronger vaccine supply and logistics systems. Vaccine 31: Supplement 2 B73-B80. [View]
- Eric Osei, Mohammed Ibrahim, and Gregory Kofi Amenuvegbe (2019) Effective Vaccine Management: The Case of a Rural District in Ghana”, Advances in Preventive Medicine, Volume 2019, Article ID 5287287. [View]
- Shahriar Tanvir Alama, Sayem Ahmed, Syed Mithun Ali, Sudipa Sarker, Golam Kabir, and Asif ul- Islam (2021) Challenges to COVID-19 vaccine supply chain: Implications for sustainable development goals, Int Int J Prod Econ 239: 108193. [View]
Article Type
Research Article
Publication history
Received: March 31, 2022
Accepted: April 12, 2022
Published: April 19, 2022
Citation:
Lim H, Wong S, Zheng Yi, Sun P, Yang XT, Cheng CH (2022) Vaccine Order, Distribution, Tracking, and Monitoring in Hong Kong Vaccination Program. Med Case Rep Ther Stud 03(02): 106–122.
Hanah Lim, Simon Wong*, Yi Zheng, Peng Sun, XinTao Yang, and Chun Hung Cheng
Logistics and Supply Chain MultiTech R&D Centre, Level 11, Cyberport 2, 100 Cyberport Road, Hong Kong SAR
*Corresponding author
Simon Wong,
Logistics and Supply Chain MultiTech R&D Centre,
Level 11, Cyberport 2,
100 Cyberport Road, Hong Kong, SAR;