- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Spontaneous Pneumomediastinum and COVID-19 Pneumonia: An Intensive Challenge
- Home
- Back to Journal
- Article Details
Introduction
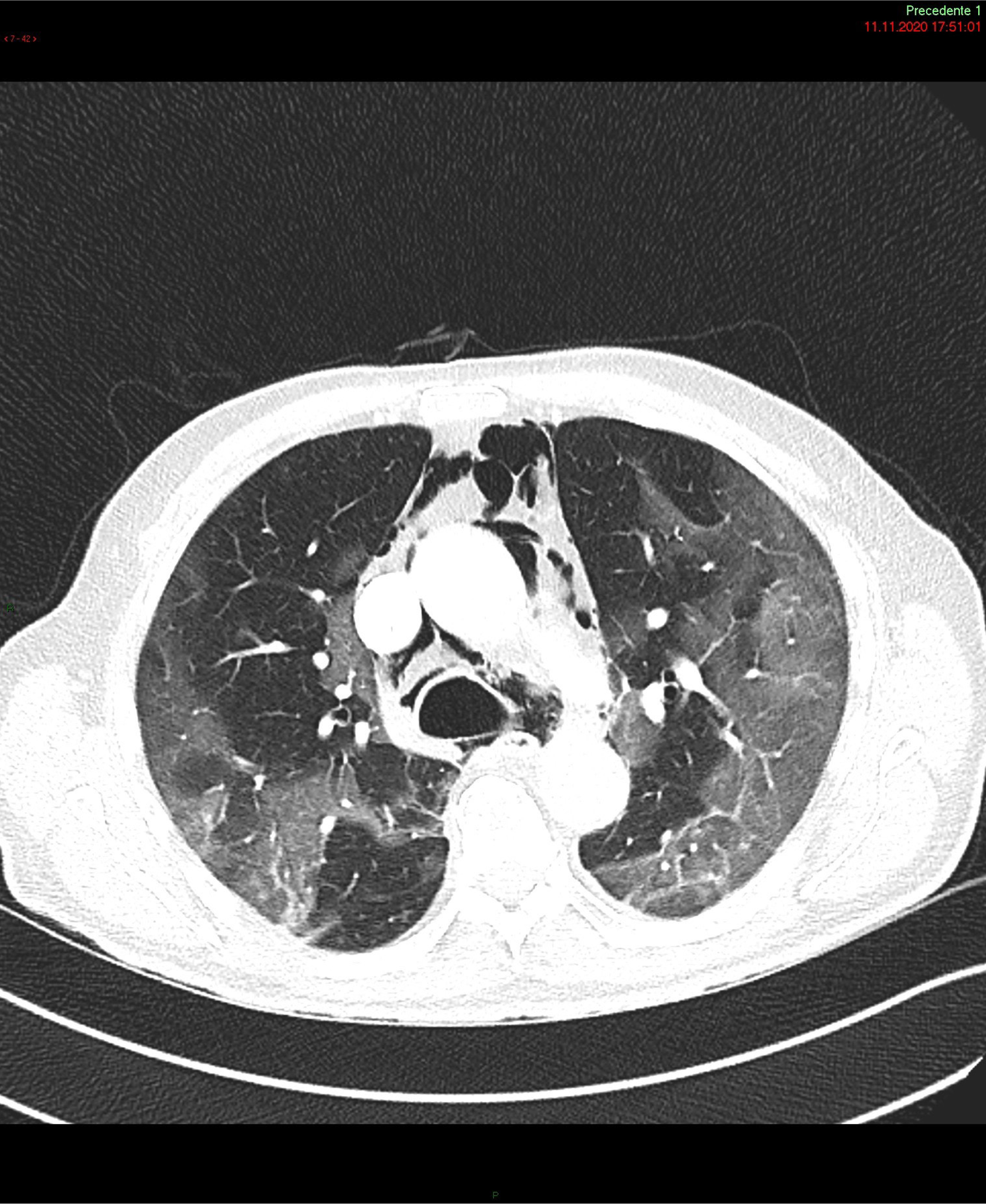
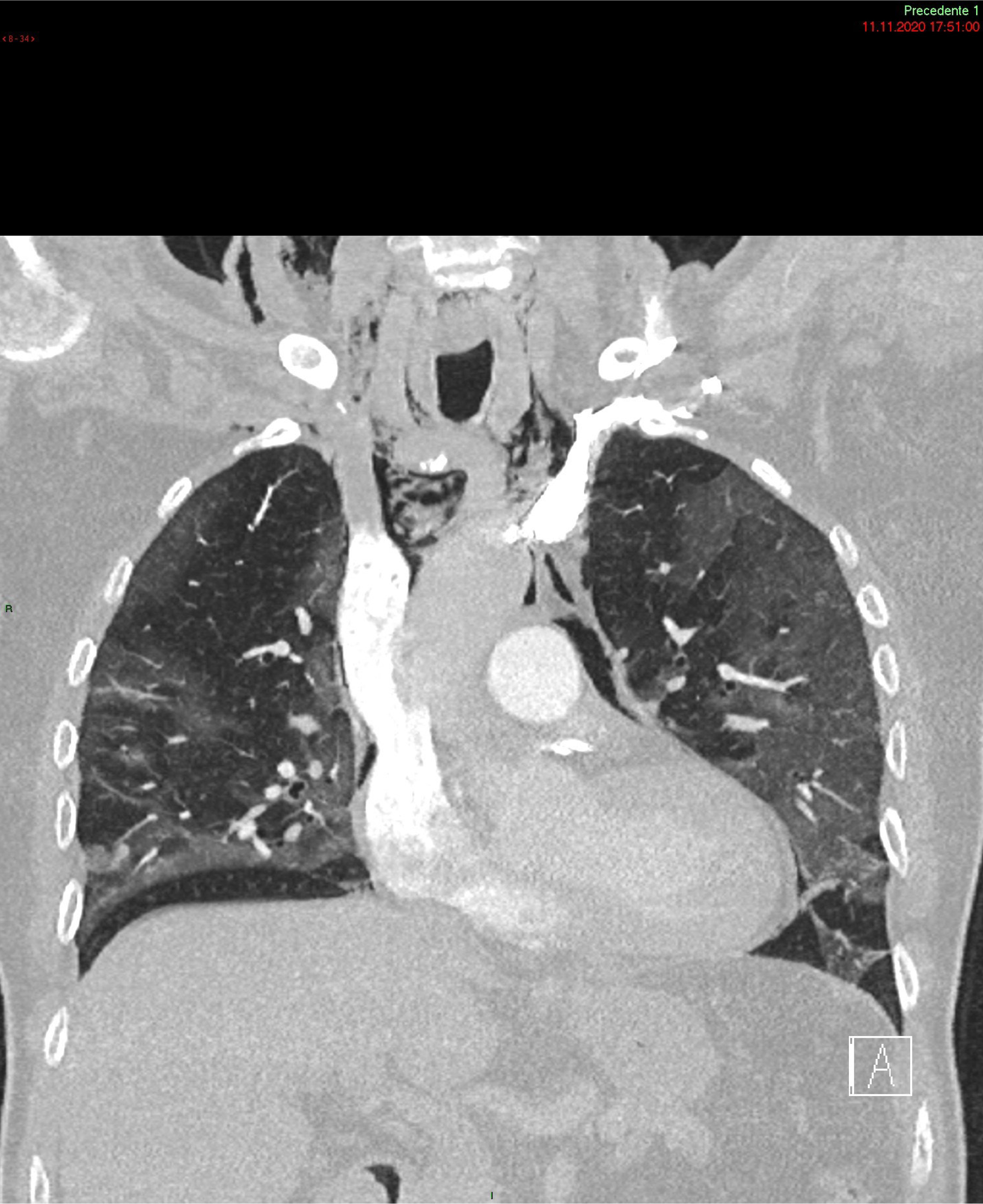
A 72-year-old patient was admitted with a story of 10 days of fever, shortness of breath, cough and diarrhea, resulting positive to SARS-CoV-2 infection, requiring oxygen at 3 L/min to reach a SpO2 of 92%. Laboratory analysis presented LDH 505 U/L, Ferritine 1674 ng/ml, CRP 339 mg/l, WBC 13.0 G/L, D-dimer 581 ng FEU/ml. A treatment with Dexamethasone 6 mg die and intravenous Remdesevir were administered. During the hospitalization, patient’s conditions worsened; an angiographic CT scan was performed showing pneumatosis extending from the mediastinal pericardium to the chest wall and the cervical soft tissues to the right side. No signs for pneumothorax or pulmonary embolism were identified. The patient presented a pneumomediastinum without clear etiology.
The Topic
Spontaneous Pneumomediastinum (SP) is defined as the presence of air collection surrounding mediastinal structures further spreading into the pleural, pericardial and peritoneal spaces or into the soft tissue of the neck and chest wall. It can be secondary to severe blunt chest trauma (10% of cases), esophageal or tracheobronchial lesions (<2% of cases) [1] or barotrauma due to Mechanical Ventilation (MV). As a diagnosis of exclusion, it can be defined spontaneous pneumomediastinum (SP) when unrelated to the above mentioned mechanisms [2–4].
COVID-19 pneumonia is a viral disease caused by SARS-CoV-2, affecting over 78.100.000 patients worldwide with 1.700.000 deaths until the end of December 2020 [5]. The related acute respiratory failure usually occurs in settings of a severe bilateral interstitial pneumonia, with symptoms such as fever, dyspnea and persistent cough [6–8].
SP and Subcutaneous Emphysema (SE) have been reported as uncommon clinical findings in patient with SARS- CoV-2 infection (Figure). Case series of patients with COVID-19 pneumonia and SP not on MV [9] showed that the SP was more likely to develop on 13th – 17th day since the onset of symptoms [9]. The most common associated conditions reported were older age, obesity and dyslipidemia, hypertension, diabetes mellitus, renal disease and asthma. Its occurrence is believed to be related to an increased pressure in the interstitial space, which may favor alveolar rupture hence resulting in pneumomediastinum [10–12]. Therefore, invasive MV as well as coughing, emesis, a strong Valsalva maneuver or asthma exacerbation are considered possible triggering events [1,9,13,14]. Additional predisposing factors are high-dose corticosteroid therapy and anatomical anomalies [1,4, 9].
Etiology
Respiratory infections such as S. aureus pneumonia, Cytomegalovirus and P. jirovecii pneumonia [15], bronchiolitis, rhinovirus, human bocavirus and respiratory syncytial virus [16], H1N1 infection [17], Severe Acute Respiratory Syndrome (SARS) and COVID-19 pneumonia can also cause severe diffuse alveolar damage [18]. The damage may lead to interstitial emphysema, dissection of the bronchovascular sheets and spreading of the emphysema into the mediastinum and the soft subcutaneous tissue, in a phenomenon known as “Macklin effect” [1,4,18]. Emerging autopsy reports performed in COVID-19 patients revealed that diffuse alveolar damage appears to be the predominating histopathologic pattern of lung injury in COVID-19 patients [19,20]. Increased local pressure gradient causes the dissection of interstitial tissues-air thus reaches the mediastinum, where it generates a pneumomediastinum. Air can also reach the thoracic inlet and the soft tissues of the neck, causing cervicofacial subcutaneous emphysema [21,22]. Lemmers et al. [23] observed a seven-fold increase in SP and SE rate in patients with ARDS in the setting of a COVID-19 pneumonia, requiring MV; however, this study showed that neither high tidal volumes nor respiratory pressures where significantly correlated to the development of a SP. In fact, the origin of SP and SE was rather attributed to a greater lung frailty and to Macklin effect [1]; in this scenario, all mechanism of lung damages induced by SARS-COV-2 (like cytokines burst, thrombotic diathesis, local trauma) may increase the risk of SP [19].
The Intensive Challenge
SP is usually a benign self-limiting disease when presented on its own and it often resolves with conservative treatments. However, in patients with COVID-19 pneumonia, seems to be associated to a poor prognosis and a more severe outcome [1,9]. Furthermore, some cases of SP and pneumopericardium can be complicated and can require invasive MV [24], becoming a huge challenge.
Since it could increase the risk of developing SP, MV is contraindicated in COVID-19 patients. In fact, MV is associated both to a worsening of patient’s clinical conditions and to a greater incidence of tension pneumothorax, which leads to respiratory and hemodynamic instability [26] requiring rapid invasive decompression [27]. Similarly, during the SARS outbreak in 2003, a retrospective study showed that 46% patients presented bilateral pneumothorax and most of them required bilateral chest tube insertion [25] while 38% required intubation and 31% of them died [25]. In a retrospective study including 187 patients with COVID-19, Loffi et al. as well observed that oro-tracheal intubation in patients with ARDS and SP was associated to a poor outcome, a higher incidence of complications and a high mortality rate [24].
In this scenario, conservative treatment with proper antibiotic therapy, conventional oxygen therapy and anticoagulant prophylaxis seems to be associated to better outcomes [21]. In case MV is required, an adequate sedation level to avoid coughing or ineffective respiratory efforts seems to be associated with a lower risk of SP and pneumothorax. CT scan could be useful to study both the evolution of the underlying disease at an alveolar level and to exclude a SP. To date, however, this strategy has not yet been officially validated, while SP remains as important challenge in critically ill COVID-19 patients.
Figure 1: Chest computed tomography scan: air leakage and dissection along bronchovascular sheaths with pulmonary interstitial emphysema and pneumomediastinum, widely extending along the muscle bundles of the chest and neck causing subcutaneous emphysema.
Figure 2: Chest computed tomography scan: air leakage and dissection along bronchovascular sheaths with pulmonary interstitial emphysema and pneumomediastinum, widely extending along the muscle bundles of the chest and neck causing subcutaneous emphysema.
Reference
- Wintermark M, Schnyder P (2001) The Macklin effect: A frequent etiology for pneumomediastinum in severe blunt chest trauma. Chest 120: 543–547. [View]
- Takada K, Matsumoto S, Hiramatsu T, Kojima E, Watanabe H, et al. (2008) Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med 102: 1329–1334. [View]
- Aghajanzadeh M, Dehnadi A, Ebrahimi H, Fallah Karkan M, Khajeh Jahromi S, et al. (2015) Classification and Management of Subcutaneous Emphysema: a 10-Year Experience. Indian J Surg 77: 673–677. [View]
- Kolani S, Houari N, Haloua M, Alaoui Lamrani Y, Boubbou M, et al. (2020) Spontaneous pneumomediastinum occurring in the SARS-COV-2 infection. IDCases 21: 4–7. [View]
- Johns Hopkins University & Medicine, John Hopkins University and Medicine (2020) COVID-19 Map - Johns Hopkins Coronavirus Resource Center. Johns Hopkins Coronavirus Resour Cent. [View]
- Rothan HA, Byrareddy SN (2020) The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun 109: 102–433. [View]
- CDC (2020) Symptoms of Coronavirus. Cdc [View]
- Guan W, Ni Z, Hu YYHY, Liang W, Ou C, et al. (2020) Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 382: 1708–1720. [View]
- Manna S, Maron SZ, Cedillo MA, Voutsinas N, Toussie D, et al. (2020) Spontaneous subcutaneous emphysema and pneumomediastinum in non-intubated patients with COVID-19. Clin Imaging 67: 207–213. [View]
- Kass DA, Duggal P, Cingolani O (2020) Obesity could shift severe COVID-19 disease to younger ages. Lancet 395: 1544–1545. [View]
- Zhou F, Yu T, Du R, Fan G, Liu Y, et al. (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395: 1054–1062. [View]
- Grasselli G, Zangrillo A, Zanella A, Antonelli M, Cabrini L, et al. (2020) Baseline Characteristics and Outcomes of 1591 Patients Infected with SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA - J Am Med Assoc 323: 1574– 1581. [View]
- Caceres M, Ali SZ, Braud R, Weiman D, Garrett HE (2008) Spontaneous Pneumomediastinum: A Comparative Study and Review of the Literature. Ann Thorac Surg 86: 962–966. [View]
- Sachdeva R, Sachdeva S (2015) Coughing leading to spontaneous pneumomediastinum and subcutaneous emphysema in a young male. Med J Dr DY Patil Univ 8: 344. [View]
- Park YK, Jung HC, Kim SY, Kim MY, Jo K, et al. (2014) Spontaneous pneumomediastinum, pneumopericardium, and pneumothorax with respiratory failure in a patient with AIDS and pneumocystis jirovecii pneumonia. Infect Chemother 46: 204–208. [View]
- Emiralio?lu N, Ozcan HN, O?uz B, Yalçin E, Do?ru D, et al. (2015) Pneumomediastinum, pneumorrhachis and subcutaneous emphysema associated with viral infections: Report of three cases. Pediatr Int 57: 1038–1040. [View]
- Chekkoth SM, Naga SR, Valsala N, Kumar P, Raja RS (2019) Spontaneous pneumomediastinum in H1N1 infection: Uncommon complication of a common infection. J R Coll Physicians Edinb 49: 298–300. [View]
- Xu Z, Shi L, Wang Y, Zhang J, Huang L, et al. (2020) Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med 8: 420–422. [View]
- Barton LM, Duval EJ, Stroberg E, Ghosh S, Mukhopadhyay S (2020) COVID-19 Autopsies, Oklahoma, USA. Am J Clin Pathol 153: 725–733. [View]
- Carsana L, Sonzogni A, Nasr A, Rossi RS, Pellegrinelli A, et al. (2020) Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: a two-centre descriptive study. Lancet Infect Dis 20: 1135–1140. [View]
- Singh A, Bass J, Lindner DH (2020) Rare Complication of Pneumomediastinum and Pneumopericardium in a Patient with COVID-19 Pneumonia. Case Rep Pulmonol . [View]
- Vanzo V, Bugin S, Snijders D, Bottecchia L, Storer V, Barbato A (2013) Pneumomediastinum and pneumopericardium in an 11-year-old Rugby Player: A Case Report. J Athl Train 48: 277–281. [View]
- Lemmers DHL, Abu Hilal M, Bnà C, Prezioso C, Cavallo E (2020) Pneumomediastinum and subcutaneous emphysema in COVID-19: barotrauma or lung frailty? ERJ Open Res 6: 00385–02020. [View]
- Loffi M, Regazzoni V, Sergio P, Martinelli E, Stifani I, et al. (2020) Spontaneous pneumomediastinum in COVID-19 pneumonia. Monaldi Arch Chest Dis 90: 604– 607. [View]
- Chu CM, Leung YY, Hui JYH, Hung IFN, Chan VL, et al. (2004) Spontaneous pneumomediastinum in patients with severe acute respiratory syndrome. Eur Respir J 23: 802–804. [View]
- Liew MF, Siow WT, Yau YW, See KC (2020) Safe patient transport for COVID-19. Crit Care 94. [View]
- Shennib HF, Barkun AN, Matouk E, Blundell PE (1988) Surgical decompression of a tension pneumomediastinum. A ventilatory complication of status asthmatics. Chest 93: 1301–1302. [View]
Article Type
Short Commentary
Publication history
Received: January 10, 2021
Accepted: January 20, 2021
Published: January 23, 2021
Citation:
Glotta A, Ramistella A, Depaoli R, Saporito A, Borgeat A, Ceruti S (2021) Spontaneous Pneumomediastinum and COVID-19 Pneumonia: An Intensive Challenge. Clar J Infect Dis Ther 02(02): 129–134.
Andrea GLOTTA1, Alice RAMISTELLA2, Remigio DEPAOLI3, Andrea SAPORITO4, Alain BORGEAT5, and Samuele CERUTI1*
1Department of Critical Care, Clinica Luganese Moncucco, Lugano, Switzerland
2Surgery Department, Clinica Luganese Moncucco, Lugano, Switzerland
3Radiology Division, Clinica Luganese Moncucco, Lugano, Switzerland
4Service of Anesthesiology, Ospedale Regionale di Bellinzona e Valli, 6500 Bellinzona, Switzerland
5Balgrist University Hospital, Anaesthesiology Division, Zurich, Switzerland
*Corresponding author
Samuele Ceruti,
Department of Critical Care,
Clinica Luganese Moncucco,
6900, Lugano,
Switzerland;