- 4192 Devonwood Way, Ashburn, Virginia, 20148
- Helpline: +1 (703) 665-3747
Presence of Anti-SARS-CoV-2 Antibodies of Class IgA But Not IgG in a COVID-19 Patient
- Home
- Back to Journal
- Article Details
Abstract
An infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) usually induces the formation of specific binding antibodies of class IgA and IgG. We present a case of mild coronavirus disease 2019 (COVID-19) with an atypical immune response. The patient developed only extremely weak levels of anti-SARS-CoV-2 IgG antibodies against the nucleocapsid protein (NCP) and did not develop anti-SARS-CoV-2 IgG antibodies against the surface spike glycoprotein (S1), whereas a pronounced increase in anti-SARS-CoV-2 S1 IgA antibodies was observed. Due to successive multiparametric serological testing, this case provides an opportunity to report unusual antibody kinetics as well as to demonstrate how a patient’s immune response can be analyzed effectively.
Keywords: antibodies, COVID-19, IgA, SARS-CoV-2, serological testing.
Introduction
The majority of COVID-19 patients with a confirmed PCR diagnosis suffer only a mild illness [1] and develop specific IgA and IgG antibodies against viral proteins of SARS-CoV-2 within 14 days post onset of symptoms, which remain elevated after initial viral clearance [2–5]healthy adults (101 subjects. Here, we describe a mildly symptomatic COVID-19 patient with consistently and strongly positive levels of anti-SARS-CoV-2 S1 IgA but not IgG antibodies during long-term monitoring. This report will be helpful to researchers by providing an example of how serological tests may be effectively used to reveal also uncommon characteristics of an individual’s immune response.
Case description
The patient (female, 58 years, non-smoker, regular weight, sporty fit, no comorbidities) works as a general practitioner in Germany. Through frequent contact with children suffering from respiratory infections, she has 5–10 respiratory viral infections per year. In the first week of March 2020, she had a respiratory infection with accompanying bronchitis, which was subsiding at the time of subsequent SARS-CoV-2 contact. The patient was negative for influenza virus on March 2 by PCR.
On March 8 she attended a familial get-together with her younger brother, the brother’s partner, her sister and mother. The younger brother and his partner had returned from Ischgl (Northern Tyrol, Austria) on March 7 with flu-like symptoms (subfebrile temperatures, headache and body aches). On March 10, media reported that Ischgl might be a hotspot and thus the brother called the responsible health department in their region to report their return from Ischgl and their flu-like symptoms. The health department informed them that Ischgl would not belong to the SARS-CoV-2 risk area of South Tyrol and, as they were not suspected to be COVID-19 patients, recommended influenza throat swabs. Influenza was a differential diagnosis, because the symptoms were consistent and the number of influenza cases was high in early March 2020. At the request of the patient of this case report, the brother and his partner were additionally tested for SARS-CoV-2. On March 10, throat swabs were taken for determination of both influenza virus and SARS-CoV-2 by PCR. The results were positive for SARS-CoV-2 infection. From March 12–21 the brother developed the following symptoms: pronounced coughing, fever: 39–40 degrees Celsius, and beginning pneumonia (bilateral) for three days. His partner had a slight fever for a few days. All family members went into quarantine.
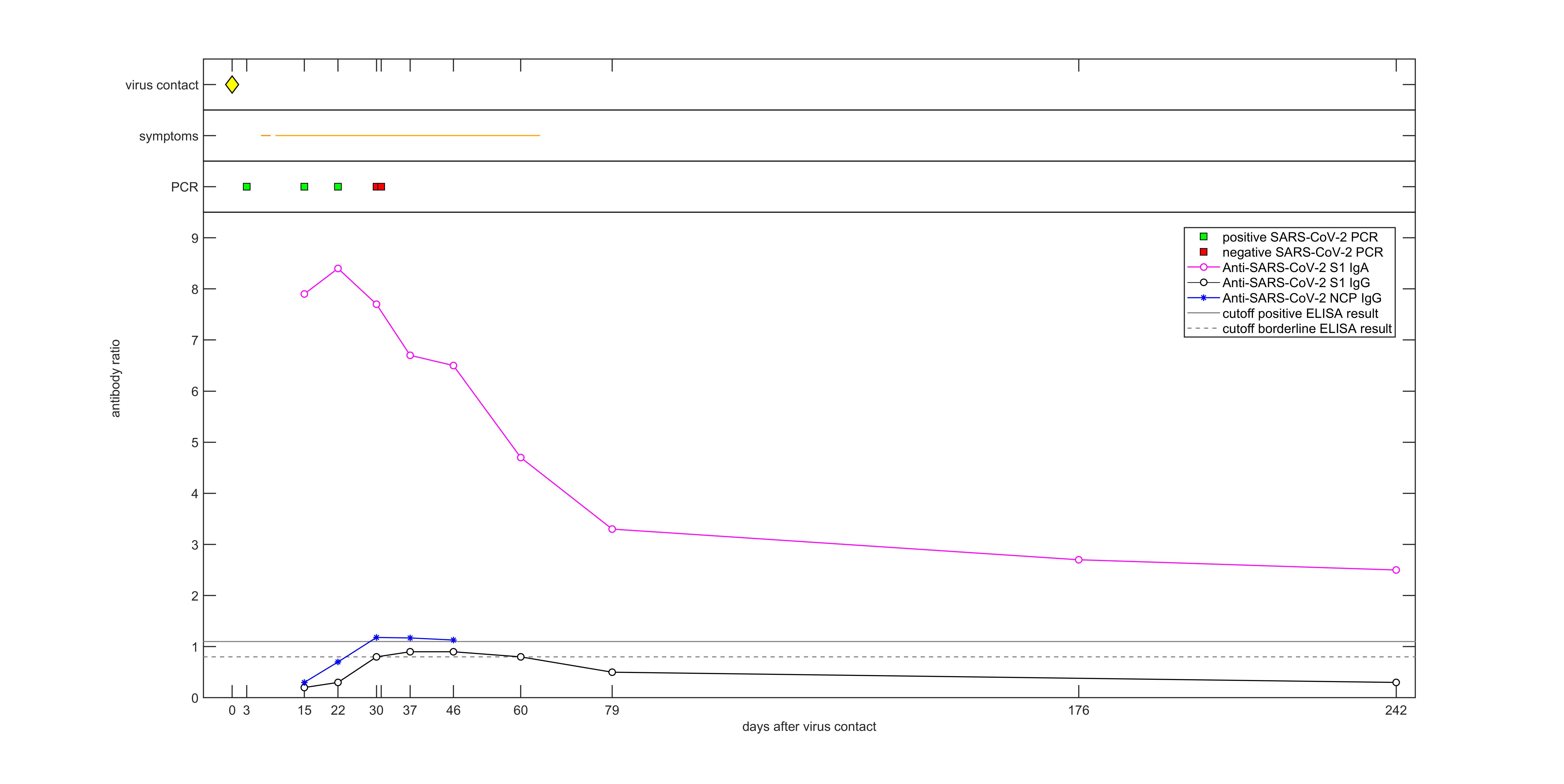
As a contact person of first degree, the patient of this case report was tested for SARS-CoV-2 by PCR based on a throat swab taken on March 11. The result was positive. She had a mild disease course (March 14–16, Figure 1), including fatigue, headache, body aches, but her body temperature stayed continuously below 37.5 degrees Celsius. Cardiac arrhythmias (supraventricular single extrasystoles, 2:1 or 3:1 coupling) occurred on March 15 and persisted, with varying intensity (initially pronounced accumulation), for about six months. The patient has had a tendency to extrasystoles since she had suffered from influenza with myocardial involvement in January 2017. Then, on March 17, she observed a sudden onset of anosmia and dysgeusia. The sense of smell and taste gradually returned after 6–8 weeks (Figure 1). However, in December 2020, its subtleties were still missing. The patient was quarantined from March 11 to April 9, as ordered by the regional health department. A throat swab taken on April 7 was negative for SARS-CoV-2 by PCR. The patient received neither medical treatment nor medication. By December 2020, the patient no longer had physical or psychological after-effects or long-term effects.
Figure 1: Timeline of virus contact, symptoms, PCR results and longitudinal titers of anti-SARS-CoV-2 antibodies against the nucleocapsid protein (NCP) and spike protein (S1).
Diagnostic assessment
Direct SARS-CoV-2 detection by PCR yielded positive results on swab samples taken on March 11, 23 and 30 (Ct-values: 30.67, 34.61, 37.73, respectively), whereas negative results were obtained for swab samples taken on April 7 and 8 (Table 1, Figure 1).
Table 1: Results of PCRs and five serological assays. Positive and borderline results of the semiquantitative assays are printed in bold and underlined, respectively. For the Anti-SARS-CoV-2 ELISAs, ratios ≥0.8 and ≥1.1 were evaluated as borderline and positive, respectively. For the recomWell ELISA, ≥20 U/ml and >24 U/ml were evaluated as borderline and positive, respectively. For the Elecsys Anti-SARS-CoV-2 NCP IgGAM ECLIA, cut-off indices (COI) <1.0 and ≥1.0 were evaluated as non-reactive and reactive, respectively.
|
Days after virus contact |
PCR SARS-CoV-2 |
PCR |
Anti-SARS-CoV-2 ELISA (IgG) [ratio] |
Anti-SARS-CoV-2 NCP ELISA (IgG) [ratio] |
Anti-SARS-CoV-2 ELISA (IgA) [ratio] |
recomWell SARS-CoV-2 NCP IgG ELISA [U/ml] |
Elecsys Anti-SARS-CoV-2 NCP IgGAM ECLIA [COI] |
|
–6 |
|
negative |
|
|
|
|
|
|
3 |
positive |
|
|
|
|
|
|
|
15 |
positive |
|
0.2 |
0.3 |
7.9 |
9.3 |
0.238 |
|
22 |
positive |
|
0.3 |
0.7 |
8.4 |
17.7 |
1.08 |
|
30 |
negative |
|
0.8 |
1.18 |
7.7 |
34.8 |
4.53 |
|
31 |
negative |
|
|
|
|
|
|
|
37 |
|
|
0.9 |
1.17 |
6.7 |
39.4 |
7.92 |
|
46 |
|
|
0.9 |
1.13 |
6.5 |
37.2 |
10.7 |
|
60 |
|
|
0.8 |
|
4.7 |
|
|
|
79 |
|
|
0.5 |
|
3.3 |
|
|
|
176 |
|
|
|
|
2.7 |
|
|
|
242 |
|
|
0.3 |
|
2.5 |
|
|
Serological analysis was performed on nine serial serum samples taken 15–242 days after virus contact using the Anti-SARS-CoV-2 ELISAs IgG and IgA (EUROIMMUN) based on the recombinantly produced S1 domain of the spike domain, including the receptor binding domain (RBD) of SARS-CoV-2, as well as the Anti-SARS-CoV-2 NCP ELISA IgG (EUROIMMUN) based on a modified nucleocapsid protein (NCP) of SARS-CoV-2. The dominant feature of this patient’s immune response was a continuously high IgA titer (Figure 1). Titers of anti-NCP IgG were marginally positive between days 30 and 46 after virus contact, while titers of anti-S1 IgG reached only borderline values between days 30 and 60 after virus contact (Table 1, Figure 1).
Antibody results were confirmed by congruent results (Table 1) obtained with the recomWell SARS-CoV-2 IgG ELISA using a highly purified recombinant nucleocapsid protein as antigen (Mikrogen) and the Elecsys Anti-SARS-CoV-2 for qualitative detection of antibodies, including IgG, IgA and IgM, to SARS-CoV-2 using a recombinant nucleocapsid protein as antigen (Roche).
The patient’s younger brother and his partner developed typical kinetics of both anti-SARS-CoV-2 S1 IgA and IgG antibodies.
T-cell activity was indirectly determined through detection of interferon gamma release by SARS-CoV-2-specific T cells stimulated by SARS-CoV-2-specific antigens using the SARS-CoV-2 Interferon Gamma Release Assay (IGRA, EUROIMMUN, for research use only). The result was 65 mIU/ml in a whole blood sample taken in January 2021.
Discussion
While it is common to see positive PCR and negative serological findings at early time points in the course of infection, this case report on an immunocompetent PCR-confirmed COVID-19 patient is unusual with regard to the continuously negative or borderline anti-S1 IgG results despite the continuous positivity for anti-S1 IgA antibodies.
IgA antibodies play an essential role in protective immunity via toxin- and viral-neutralizing activities in the respiratory and gastrointestinal tracts [6]. However, in most COVID-19 patients with mild disease severity, IgA is not considered as a marker for the long-term monitoring of an immunological response after infection [7, 8]. Current research shows that mild COVID-19 cases usually develop transiently positive anti-SARS-CoV-2 serum IgG and IgA titers (with IgG being slightly higher than IgA), whereas patients with severe COVID-19 show considerable increases in both specific serum IgA and IgG titers [9–11]. It was reported that in patients with mild disease the S1-specific IgA response may be transient and delayed [9]. This prior evidence is in stark contrast to the immune response of this patient, which is characterized by continuously high IgA titers between days 15 and 242 after virus contact. One study reported a correlation between very high titers of specific IgA antibodies and the development of acute respiratory distress syndrome, which was however not observed in this patient [9]. An upregulated IgA production may be the result of increased levels of cytokines that promote antibody switching in SARS-CoV-2 infection [12]. Potential immune mechanisms by which a pronounced IgA response may be associated with a mild severity of COVID-19 will need to be further explored.
Regarding a weak IgG response in COVID-19 patients, preliminary evidence suggests that it is associated with a significantly higher SARS-CoV-2 clearance rate [13].
It is yet unknown whether preexisting immunity to related coronaviruses is an influential factor for the humoral response to SARS-CoV-2 or COVID-19 severity. It is also unknown, how SARS-CoV-2 interacts with other respiratory viruses within a host, e.g. in case of sequential infections or coinfections. Using mathematical modelling, it was shown that SARS?CoV?2 replication is easily suppressed when initiated simultaneously or after infection with another respiratory virus [14]. A likely explanation is the lower replication rate of SARS-CoV-2 compared to influenza A virus, respiratory syncytial virus, human rhinovirus, parainfluenza virus and human metapneumovirus [14]. Hence, a possible cause for the absence of anti-SARS-CoV-2 S1 IgG antibodies in this patient might be that she had had a recent viral respiratory infection (not influenza) that might have influenced the humoral immune response to SARS-CoV-2, for instance, the coronavirus could have triggered pre-existing cellular responses through cross-reaction that may prime protective responses [15]. This may lead to unconventional patterns of immune response similar to those of secondary immune responses [15].
According to the manufacturer of the assays used here, cross-reactivities were observed in sera from patients with anti-SARS-CoV(-1) IgG antibodies but not in sera from patients with MERS-CoV, HCoV-229E, HCoV-NL63, HCoV-HKU1 or HCoV-OC43 infections.
Interestingly, in a cohort of health care workers with a history of anti-tuberculosis vaccination with Bacillus Calmette-Guérin was associated with decreased seroprevalence of anti-SARS-CoV-2 IgG and mild COVID-19 symptoms [16]. The patient reported here, however, had not been vaccinated against tuberculosis, but a Mendel-Mantoux tuberculin skin test conducted several years ago had been positive. The patient might have had a mild COVID-19 course due to a prior infection with Mycobacterium tuberculosis bacteria that boosted the innate immune response to SARS-CoV-2 [17].
Thus far, possible protective factors that might have contributed to the observed partial response of the immune system in this patient remain speculative.
The spike protein of coronaviruses, especially the RBD, is able to induce neutralizing antibodies and T-cell immune responses [18] IgG antibodies against the S1 subunit of the SARS-CoV-2 spike protein and specific long-lived T cells are suspected to play the most relevant roles in virus neutralization and sustained immunity. An implication of the absence of anti-SARS-CoV-2 S1 IgG antibodies may be the possibility that protective antibodies did not develop and hence a protection against re-infection cannot be assumed [19–21].
Interferon gamma is an important signaling molecule of the immune system which is released by antigen-specific T cells upon contact with a pathogen. Its detection can support evaluating the cellular immune response in SARS-CoV-2 infected (or vaccinated) individuals. In January 2021, the patient’s T cell activity against SARS-CoV-2 was very low. Since evidence on the durability of the interferon gamma response to SARS-CoV-2 is scarce [22], an interpretation of this observation should be exercised with caution. Low levels of interferon gamma produced by SARS-CoV-2 reactive T cells might be a result of cross-reactivity due to previous encounters with endemic coronaviruses, as reported by Weiskopf et al and Braun et al, who observed this in sera from SARS-CoV-2-naive healthy donors [23, 24]
The main applications of serological testing during the current pandemic are seroprevalence studies, the supplementation of primary diagnosis and monitoring of the immune response of vaccinees. Detection of IgG antibodies against SARS-CoV-2 plays the predominant role in serological testing, although an IgG antibody response does not develop reliably in all infected individuals [7, 25]. A clear understanding of how serological assays can be used effectively will support ongoing public health efforts [26]. Especially when monitoring a patient following an acute infection confirmed by direct virus detection or when serological assessment is used to support the diagnosis in a suspected (asymptomatic) case it is crucial to design the testing strategy so that it captures not only the most frequently observed antibody responses, but also includes deviant variants. Therefore, testing strategies should include detection of anti-SARS-CoV-2 IgA antibodies to support most accurate serodiagnosis of SARS-CoV-2 infection.
Highlights / Learning points
- During the current pandemic, the absence of specific IgG antibodies does not exclude previous infection with SARS-CoV-2.
- Especially in patients with mild disease severity or asymptomatic courses without PCR result, SARS-CoV-2 infection should be confirmed by testing for both IgG and IgA antibodies against SARS-CoV-2.
- The investigation of anti-SARS-CoV-2 IgA antibodies can be valuable for monitoring of COVID-19 patients.
Patient perspective
“The relatively long quarantine of 34 days was accompanied by a certain sense of unreality, a feeling of having been torn from the busy routine (and fast pace) of my everyday life just from one moment to the next. SARS-CoV-2, omnipresent in the media, had suddenly come really close due to my own infection and those of my two brothers and my younger brother’s partner. The first days were filled with worrying about my younger brother (healthy, active and without risk factors), who was having a much more severe course than myself, and also about my mother of 85 years, who lives in the same house as my brother and was just recovering from serious surgery. I first found it hard to grasp my own COVID-19 illness due to its mild course. Only when I suddenly experienced the complete loss of my olfactory sense and taste, which was very intriguing, and with the frequent extrasystoles occurring at the same time, I started having moments of fear. This was also because I was medically caring for my brother, seeing the much more severe course the infection took in his case. At the same time, my mother, who had just been released from hospital, developed a fever which lasted over days, showing significantly increased infection markers in laboratory testing (two SARS-CoV-2 PCRs, however, were negative, thank God). For around a week, we all felt like in the film Groundhog Day until my brother and mother’s fever finally broke and they started to feel better. A very gratifying experience was to feel the support, the empathy and good wishes coming from family, friends, practice staff, colleagues and patients (my illness led to more than 60 families being quarantined as patient contacts and my practice being closed for two weeks). I was very relieved that I didn’t infect any of my contact persons, and neither my two sons who live with me. That worry had been a real burden to me! On the other hand, this involuntary full brake in my life gave me precious moments of calm and reflection and made me realize once more how fragile life can be. I am very grateful and relieved that my COVID-19 illness was mild and that my family has fully recovered. I feel with all those who are making or have made a different experience. Due to my job as a general practitioner, I am still confronted with SARS-CoV-2 and COVID-19 every day and I consider myself lucky to live in a country with democratic decision-making processes, civil rights and a very good healthcare system. Many people who have to cope with COVID-19 or other severe diseases do not have this privilege.”
References
- Wu Z, McGoogan JM (2020) Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72?314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 323: 1239–1242. [View]
- Gorse GJ, Donovan MM and Patel GB (2020) Antibodies to coronaviruses are higher in older compared with younger adults and binding antibodies are more sensitive than neutralizing antibodies in identifying coronavirus-associated illnesses. J Med Virol 92: 512–517. [View]
- Okba NMA, Müller MA, Li W, Wang C, GeurtsvanKessel CH, Corman VM, Lamers MM, Sikkema RS, Bruin E de, Chandler FD, et al. (2020) Severe Acute Respiratory Syndrome Coronavirus 2 - Specific Antibody Responses in Coronavirus Disease 2019 Patients. Emerg Infect Dis 26: 1478–1488 [View]
- Long Q-X, Liu B-Z, Deng H-J, Wu G-C, Deng K, Chen Y, Liao P, Qiu J-F, Lin Y, Cai X-F, et al. (2020) Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat Med 26: 845–848. [View]
- Prévost J, Gasser R, Beaudoin-Bussières G, Richard J, Duerr R, Laumaea A, Anand SP, Goyette G, Benlarbi M, Ding S, et al. (2020) Cross-Sectional Evaluation of Humoral Responses against SARS-CoV-2 Spike. Cell reports Med 1: 100126. [View]
- Janeway CJ, Travers P, Walport M and Al. E. “The distribution and functions of immunoglobulin isotypes,” in Immunobiology: The Immune System in Health and Disease. (New York, NY: Garland Science). Available at: https://www.ncbi.nlm.nih.gov/books/NBK27100/%0A
- Fill Malfertheiner S, Brandstetter S, Roth S, Harner S, Buntrock-Döpke H, Toncheva AA, Borchers N, Gruber R, Ambrosch A, Kabesch M, et al. (2020) Immune response to SARS-CoV-2 in health care workers following a COVID-19 outbreak: A prospective longitudinal study. J Clin Virol 130: 104575. [View]
- Brandstetter S, Roth S, Harner S, Toncheva AA, Borchers N, Gruber R, Ambrosch A and Kabesch M. (2020) Symptoms and immunoglobulin development in hospital staff exposed to a SARS-CoV-2 outbreak. Pediatr Allergy Immunol 31: 841–847. [View]
- Cervia C, Nilsson J, Zurbuchen Y, Valaperti A, Schreiner J, Wolfensberger A, Raeber M, Adamo S, Emmenegger M, Hasler S, et al. (2020) Systemic and mucosal antibody secretion specific to SARS-CoV-2 during mild versus severe COVID-19. J Allergy Clin Immunol [View]
- Ibarrondo FJ, Fulcher JA, Goodman-Meza D, Elliott J, Hofmann C, Hausner MA, Ferbas KG, Tobin NH, Aldrovandi GM and Yang OO (2020) Rapid Decay of Anti–SARS-CoV-2 Antibodies in Persons with Mild Covid-19. N Engl J Med 383: 1085–1087. [View]
- Kutsuna S, Asai Y and Matsunaga A (2020) Loss of Anti–SARS-CoV-2 Antibodies in Mild Covid-19. N Engl J Med 383: 1694–1696. [View]
- Yu HQ, Sun BQ, Fang ZF, Zhao JC, Liu XY, Li YM, Sun XZ, Liang HF, Zhong B, Huang ZF, et al. (2020) Distinct features of SARS-CoV-2-specific IgA response in COVID-19 patients. Eur Respir J 56: 7–10. [View]
- Tan W, Lu Y, Zhang J, Wang J, Dan Y, Tan Z, He X, Qian C, Sun Q, Hu Q, et al. (2020) Viral Kinetics and Antibody Responses in Patients with COVID-19. medRxiv [View]
- Pinky L and Dobrovolny HM (2020) SARS-CoV-2 coinfections: Could influenza and the common cold be beneficial? J Med Virol 92: 2623–2630. [View]
- Beretta A, Cranage M and Zipeto D (2020) Is Cross-Reactive Immunity Triggering COVID-19 Immunopathogenesis? Front Immunol 11: 1–9. [View]
- Noval Rivas M, Ebinger JE, Wu M, Sun N, Braun J, Sobhani K, Van Eyk JE, Cheng S and Arditi M. (2020) BCG vaccination history associates with decreased SARS-CoV-2 seroprevalence across a diverse cohort of healthcare workers. J Clin Invest 31: e145157. [View]
- Netea MG, Giamarellos-Bourboulis EJ, Domínguez-Andrés J, Curtis N, van Crevel R, van de Veerdonk FL and Bonten M (2020) Trained Immunity: a Tool for Reducing Susceptibility to and the Severity of SARS-CoV-2 Infection. Cell 181:969–977. [View]
- Dong Y, Dai T, Wei Y, Zhang L, Zheng M and Zhou F (2020) A systematic review of SARS-CoV-2 vaccine candidates. Signal Transduct Target Ther 5: 237 [View]
- Riva E, Sainaghi PP, Turriziani O, Antonelli G, Patti G (2020) SARS-CoV-2 infection: diagnostic testing results occasionally require special attention. Emerg Microbes Infect 9: 1955–1957. [View]
- Rijkers G, Murk J-L, Wintermans B, van Looy B, van den Berge M, Veenemans J, Stohr J, Reusken C, van der Pol P and Reimerink J (2020) Differences in Antibody Kinetics and Functionality Between Severe and Mild Severe Acute Respiratory Syndrome Coronavirus 2 Infections. J Infect Dis 222: 1265–1269. [View]
- Papachristodoulou E, Kakoullis L, Parperis K and Panos G (2020) Long-term and herd immunity against SARS-CoV-2: implications from current and past knowledge. Pathog Dis 78: ftaa025. [View]
- Dan JM, Mateus J, Kato Y, Hastie KM, Faliti CE, Ramirez SI, Frazier A, Yu ED, Grifoni A, Rawlings SA, et al. (2021) Immunological memory to SARS-CoV-2 assessed for greater than six months after infection. Science (80- ) 4063: [View]
- Weiskopf D, Schmitz KS, Raadsen MP, Grifoni A, Okba NMA, Endeman H, van den Akker JPC, Molenkamp R, Koopmans MPG, van Gorp ECM, et al. (2020) Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome. Sci Immunol 5: 1–14. [View]
- Braun J, Loyal L, Frentsch M, Wendisch D, Georg P, Kurth F, Hippenstiel S, Dingeldey M, Kruse B, Fauchere F, et al. (2020) SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19. Nature 587: 270–274. [View]
- Marra A, Generali D, Zagami P, Cervoni V, Gandini S, Venturini S, Morganti S, Passerini R, Orecchia R and Curigliano G (2020) Seroconversion in patients with cancer and oncology health care workers infected by SARS-CoV-2. Ann Oncol 32: 113–119. [View]
- Theel ES, Slev P, Wheeler S, Couturier MR, Wong SJ and Kadkhoda K (2020) The Role of Antibody Testing for SARS-CoV-2: Is There One? J Clin Microbiol 1–7. [View]
Article Type
Case Report
Publication history
Received: October 12, 2022
Accepted: October 25, 2022
Published: November 07, 2022
Citation:
Nadja E, Renate F, Julia Maria K, Borchardt-Lohölter V and Claudia S (2022) Presence of anti-SARS-CoV-2 antibodies of class IgA but not IgG in a COVID-19 patient. Med Case Rep Ther Stud 03(02): 95–101.
Nadja Ewers1, Renate Froese1, Julia Maria Klemens2, Viola Borchardt-Lohölter2*, Claudia Speckbacher1
1Medical supply center DIAMEDIS Diagnostic Medicine Sennestadt GmbH, Dunlopstraße 50, 33689 Bielefeld, Germany
2Institute for Experimental Immunology, affiliated to EUROIMMUN Medizinische Labordiagnostika AG, Seekamp 31, 23560 Lübeck, Germany
*Corresponding author
Viola Borchardt-Lohölter
EUROIMMUN Medizinische Labordiagnostika AG,
Seekamp 31,
23560 Lübeck, Germany;
Phone: +49 451 2032 1619;
Fax: +49 451 2032 100;